
The department
The Department of Medicinal Chemistry is one of three departments at the Faculty of Pharmaceutical Sciences (PHARMA) at University of Copenhagen. The Faculty was an independent organization until 2006, known as the Danish University of Pharmaceutical Sciences and before that the Royal Danish School of Pharmacy. In 2007 the University merged with University of Copenhagen and the Royal Veterinary & Agricultural University to form the largest University in Scandinavia. One of the objectives of the merger has been to form a strong Health and Life Sciences cluster.
The mission of the Department of Medicinal Chemistry is to advance pharmaceutical sciences by performing drug-related research and research-based teaching within a range of topics including medicinal chemistry, natural products research & pharmacognosy, and biostructural research. The Department of Medicinal Chemistry encompasses a range of research projects directed towards discovery, design and development of model compounds and new potential drugs, discovery and characterization of novel macromolecular targets, and studies of their interactions at the molecular and cellular level. It is also engaged in the development of the underlying research methods and technologies.
The three major research areas of the department are:
- Natural products-based discovery and characterization of new chemical entities as potential new drugs.
- Discovery and characterization of novel macromolecular drug targets.
- Medicinal chemistry research towards development of potential drugs and structure-based design.
The department has several medicinal chemistry research programs integrating organic synthesis, natural products research, computational chemistry, molecular modelling, molecular pharmacology, chemical biology, recombinant protein technologies and protein crystallography, with innovative work in areas where traditional scientific disciplines intersect.
Head of the group
Professor Povl Krogsgaard-Larsen (1941) is head of the medicinal chemistry research group at the Department of Medicinal Chemistry at PHARMA. Professor Krogsgaard-Larsen initiated his research career at The Royal Danish School of Pharmacy in 1967, and has just celebrated his 40th anniversary at the institution.
In 1970, after having finished a PhD in natural products, Professor Krogsgaard-Larsen decided to introduce medicinal chemistry to the Department and to start projects on re-design of toxic natural products with the goal of developing specific research tools and compounds of therapeutic interest. He decided to explore muscimol and ibotenic acid, which is toxic constituents of the mushroom Amanita muscaria, as lead structures in drug design projects. This lead to a number of exciting findings as described below, and Professor Krogsgaard-Larsen has published ca. 450 papers related to these areas. He is also editor of a textbook in Medicinal Chemistry Textbook of Drug Design and Discovery which is widely used in teaching at all levels of medicinal chemistry.
Professor Krogsgaard-Larsen has received a wealth of acknowledgments for his accomplishments, such as the The Lundbeck Foundation Prize (1989), The Paul Ehrlich Prize (1989), The Astra Award (1991), The W. Th. Nauta Award (1996) and . The Pharmaceutical Research Achievement Award (2004). In 1998 he became the European Editor for Journal of Medicinal Chemistry. In addition to his research achievements, Professor Krogsgaard-Larsen has been a pioneer in structuring and managing a number of interdisciplinary research initiatives such as The Biotechnological Drug Research Center, PharmaBiotec, the Graduate School of Drug Research and the Drug Research Academy, all based at The Royal Danish School of Pharmacy.
In 2001 he became rector of The Royal Danish School of Pharmacy and initiated a modernization and reorganization of the institution, and one of the outcomes was the change of name to the Danish University of Pharmaceutical Sciences. He only held this position for almost two years, as he was elected as chairman of the board of directors of the Carlsberg Foundation in 2003, a position that he currently holds along with his position as professor at the Department.
Key discoveries of the group
As mentioned before, the group of Povl Krogsgaard-Larsen originated from a drug research program in the early 70es using the natural product muscimol from fly agaric mushroom as a lead structure for a number of novel analogues of the primary inhibitory neurotransmitter ã-aminobutyric acid (GABA). Later, ibotenic acid from this mushroom was employed as a lead structure for the primary excitatory neurotransmitter glutamate.
Two conformationally constrained muscimol analogues named THPO and THIP, which have bicyclic structures, turned out to be of key importance as fundamental pharmacological tools in studies of the GABA transmitter system. Whereas THPO was shown to be a specific inhibitor of GABA uptake, the closely related compound THIP specifically and potently activated GABAA receptors.
Thus, for the first time it became possible to study the pharmacology of GABA uptake mechanisms and GABAA receptor activation specifically. Using THPO as lead, PKL subsequently predicted and proved that the related monocyclic amino acids nipecotic acid and guvacine were specific and very potent GABA uptake inhibitors, and they are now standard GABA uptake inhibitors.
Nipecotic acid was further developed into Tiagabine by the drug company Novo Nordisk, Copenhagen, and is marketed as an antiepileptic agent.
THIP has been shown to activate selectively and very potently extrasynaptically located GABAA receptors and interestingly, these receptors showing distinct subunit combination are not sensitive to benzodiazepines.
It was calculated and subsequently proved by PKL that THIP would be capable of penetrating the blood-brain barrier in spite of its zwitterionic structure. THIP was shown to be a very potent non-opioid analgesic in man. In addition, THIP was shown to be a compound of low toxicity and in collaboration with the drug company H. Lundbeck, Copenhagen, THIP went to advanced phase III clinical studies under the company name Gaboxadol.
Ibotenic acid is an analogue of Glu, which is the major excitatory neurotransmitter in the CNS. The compound was transformed into the structurally related AMPA, which specifically activates a subgroup of glutamate receptors. This particular subgroup of receptors has been named AMPA receptors after the compound. The impact of this compound is underlined by the 10.000 hits a PubMed search on AMPA yields. Since then a number of AMPA analogues have been made, and several have shown interesting pharmacological properties, such as ATPA (iGluR5 selective agonist) and homo-AMPA (the only selective mGluR6 ligand).
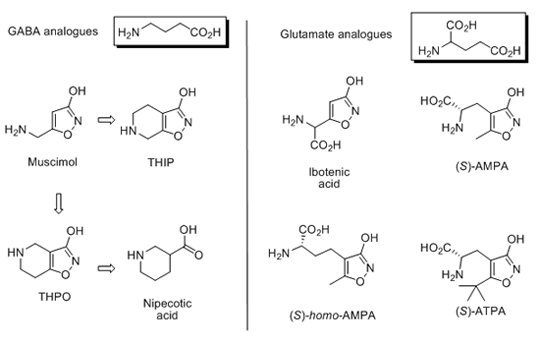
Figure 1. A selection of key pharmacological compounds affecting GABAergic and glutamatergic neurotransmission in a specific manner developed by Professor Povl Krogsgaard-Larsen.
Research Focus
Today, the group consists of three groups covering the research areas: molecular pharmacology, chemical biology and medicinal chemistry.
In general, the research focuses on neurotransmitters and their receptors and transporters. A combination of chemical, computational and pharmacological approaches is used in an integrated and interdisciplinary fashion to study neurotransmitters and their targets at the molecular level. Although GABAergic and glutamatergic neurotransmission are of primary interest other areas have caught attention within recent years also comprising studies of acetylcholine and glycine receptors, γ-hydroxybutyric acid (GHB) and its putative receptor, as well as various orphan receptors.
The research activities are still ongoing on design and synthesis of novel ligands for neurotransmitter receptors and transporters but the increasing structural knowledge on the proteins has shifted focus to a structure-based approach which involves strong collaboration with the biostructural group at the department covering molecular modeling and protein x-ray crystallographic studies. Thus, modeling is an important tool in the design of ligands but also in the design of point mutation studies. For example, highly integrated mutational studies on glutamate receptors and serotonin transporters have been completed, where both compounds and target proteins are complementary altered/mutated to give information on the recognition of ligands and activation mechanisms.
People
Professor Povl Krogsgaard-Larsen is heading the medicinal chemistry research group, in close collaboration with five associate professors, Lennart Bunch, Rasmus P. Clausen, Tommy N. Johansen and Bente Frølund, who are all working on various aspects of structure-based drug design. In addition, the Head of Department, associate professor Ulf Madsen is also a member of the team. Moreover, the group is very closely associated to the Molecular Pharmacology group and the Chemical Biology group headed by Professors Hans Bräuner-Osborne and Kristian Strømgaard, respectively.
The three groups currently employ two senior researchers, five post docs, ca. 15 PhD students, ca. 3 research assistants and ca. 5 laboratory technicians. Moreover, between 15-20 MSc students carry out their projects in these groups each year.
Selected publications
Krogsgaard-Larsen, P., Johnston, G.A., Lodge, D., Curtis, D.R. A new class of GABA agonist. Nature, 1977, 268, 53-55.
Krogsgaard-Larsen, P., Honoré, T., Hansen, J.J., Curtis, D.R., Lodge, D. New class of glutamate agonist structurally related to ibotenic acid. Nature. 1980, 284, 64-66.
Bräuner-Osborne, H., Egebjerg, J., Nielsen, E.Ø., Madsen, U, Krogsgaard-Larsen P. Ligands for glutamate receptors: design and therapeutic prospects. J. Med. Chem. 2000, 43, 2609-2645.
Information and contact
http://www.farma.ku.dk/index.php/Department-of-Medicinal-Chemis/161/0/
e-mail: pkl@carlsbergfondet.dk

Figure 2. Professor Povl Krogsgaard-Larsen at his 40th anniversary in September 2007 at Department of Medicinal Chemistry at the Faculty of Pharmaceutical Sciences, University of Copenhagen.







